|
Modelling the time–kill curves provided possibilities for optimizing antibacterial activity by increasing doses of tigecycline and/or including a loading dose for polymyxin B. 18 described the lack of sustained synergy, which was observed initially between polymyxin B and tigecycline, when using current recommended doses. Pharmacokinetic-pharmacodynamic (PKPD) modelling of in vitro experiments may better describe the interaction between two agents over time compared with chequerboard experiments, hence improving the prediction of their combined antimicrobial activity. Although synergy in chequerboards may indicate a useful mechanism, ensuring this effect persists at clinically achievable concentrations is crucial to predicting clinical utility. Most commonly, chequerboard assays, which are based on determinations of MIC with or without exposure to the second agent, 14, 15 are used. This might be due, in part, to the overly simplistic way in which synergy is defined. 13 citing strong synergistic antibacterial effect. This is in stark contrast to a meta-analysis of in vitro studies of polymyxin/carbapenem combinations by Zusman et al. baumannii based on a large multicentre randomized controlled trial. 12 concluded that there was a lack of evidence for the addition of meropenem to colistin for the treatment of carbapenem-resistant A. colistin/rifampicin and colistin/tigecycline) despite synergy in vitro cited by many sources. 10 has concluded that current evidence does not advocate for the use of previously described colistin combinations (e.g. In an effort to investigate colistin susceptibility, as well as incorporate clinical dosing regimens to better predict clinical outcome, pharmacometric modelling on time-course experiments has been developed and explored in recent years. baumannii isolates and has been thought to contribute to therapeutic failure with monotherapy, as heteroresistant isolates may appear to be susceptible to colistin in vitro by conventional susceptibility testing methods used in routine clinical laboratories (e.g. 4–6 Colistin heteroresistance is a well-described phenomenon amongst A. 4 Moreover, there have been reports of rapid evolution of colistin resistance during colistin monotherapy resulting in therapeutic failure. 4 This approach appears to present a number of advantages: relative increase in antimicrobial activity of the combination potential reduction of the dose of colistin (thereby minimizing toxicity) retardation of the development of resistance and enhancement of bactericidal activity and hence a cure for an infection that might not otherwise be curable with current monotherapies. 3 In attempts to preserve the usefulness of colistin clinically, researchers have been investigating its use in combination with other licensed antimicrobial agents. 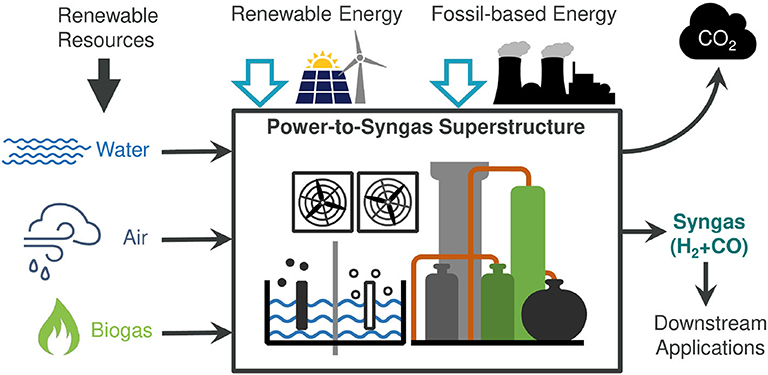
2Ĭolistin, first discovered in the 1940s and subsequently abandoned in clinical use for less toxic alternatives, is now increasingly used as an agent of last resort to combat MDR Gram-negative infections. 
1 MDR Acinetobacter baumannii (MDRAB) is a prime example of a threat to modern medicine, with its predilection for the critically ill, affecting the success and progress of a wide range of medical fields including surgery and oncology. The rise of antimicrobial resistance has captured global attention with particular focus on MDR infections for which viable therapeutic options range from few to none.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
